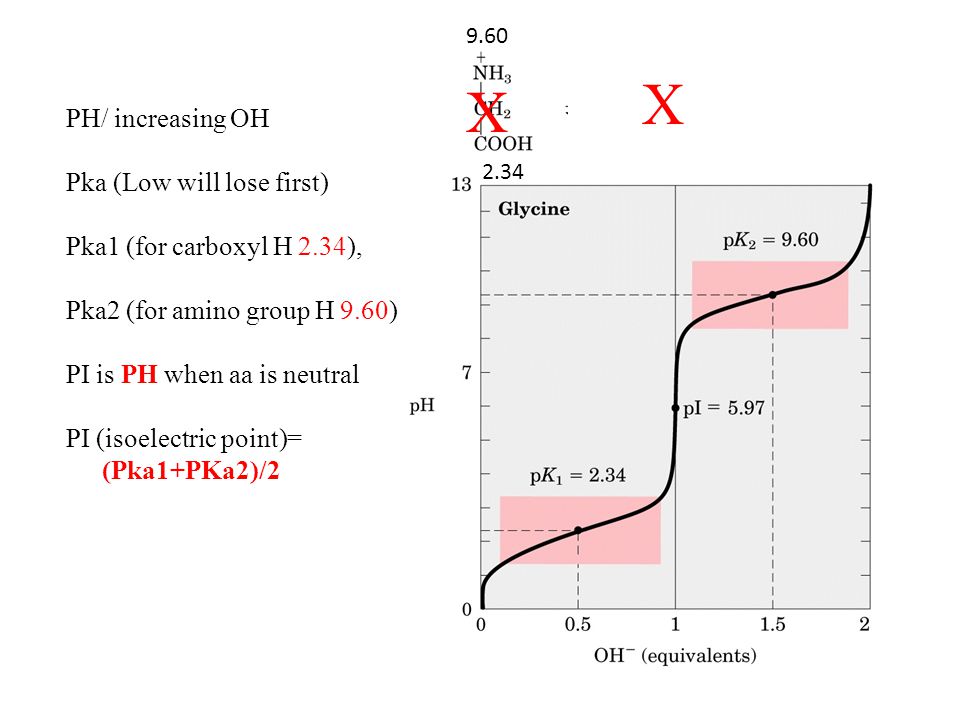
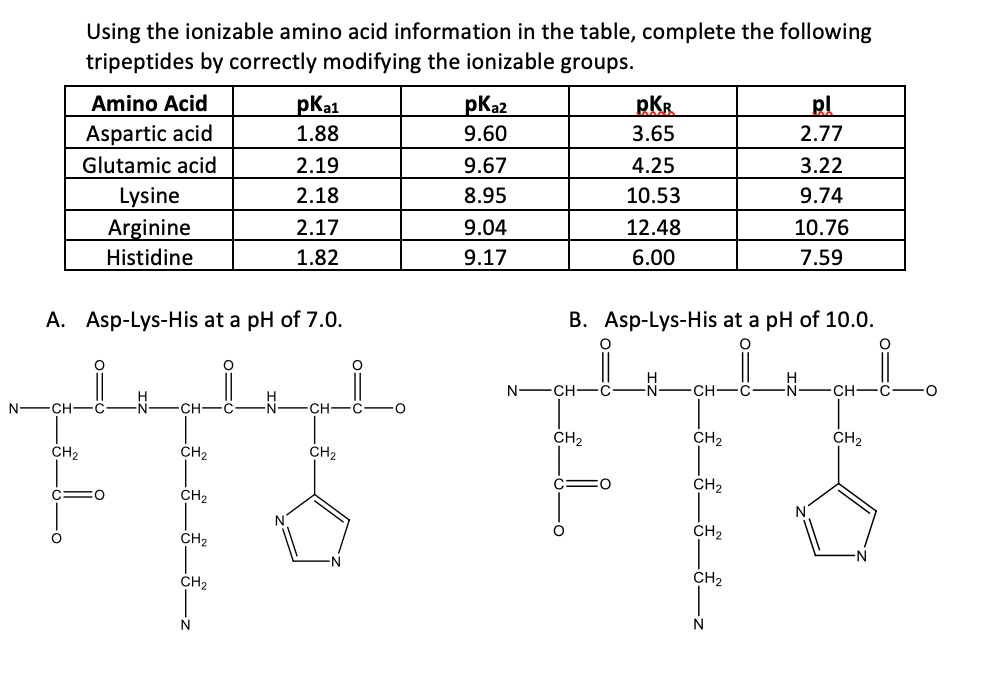
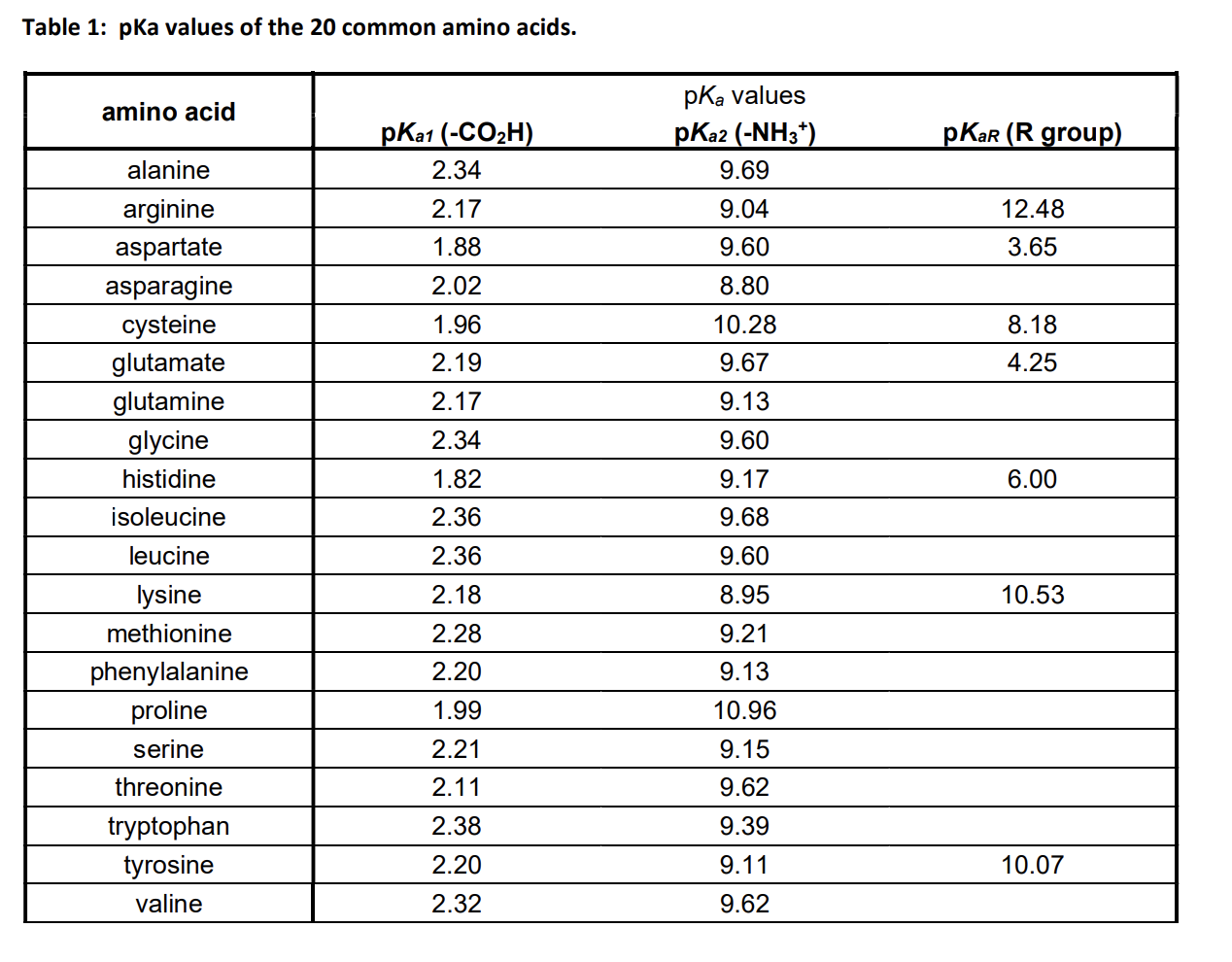
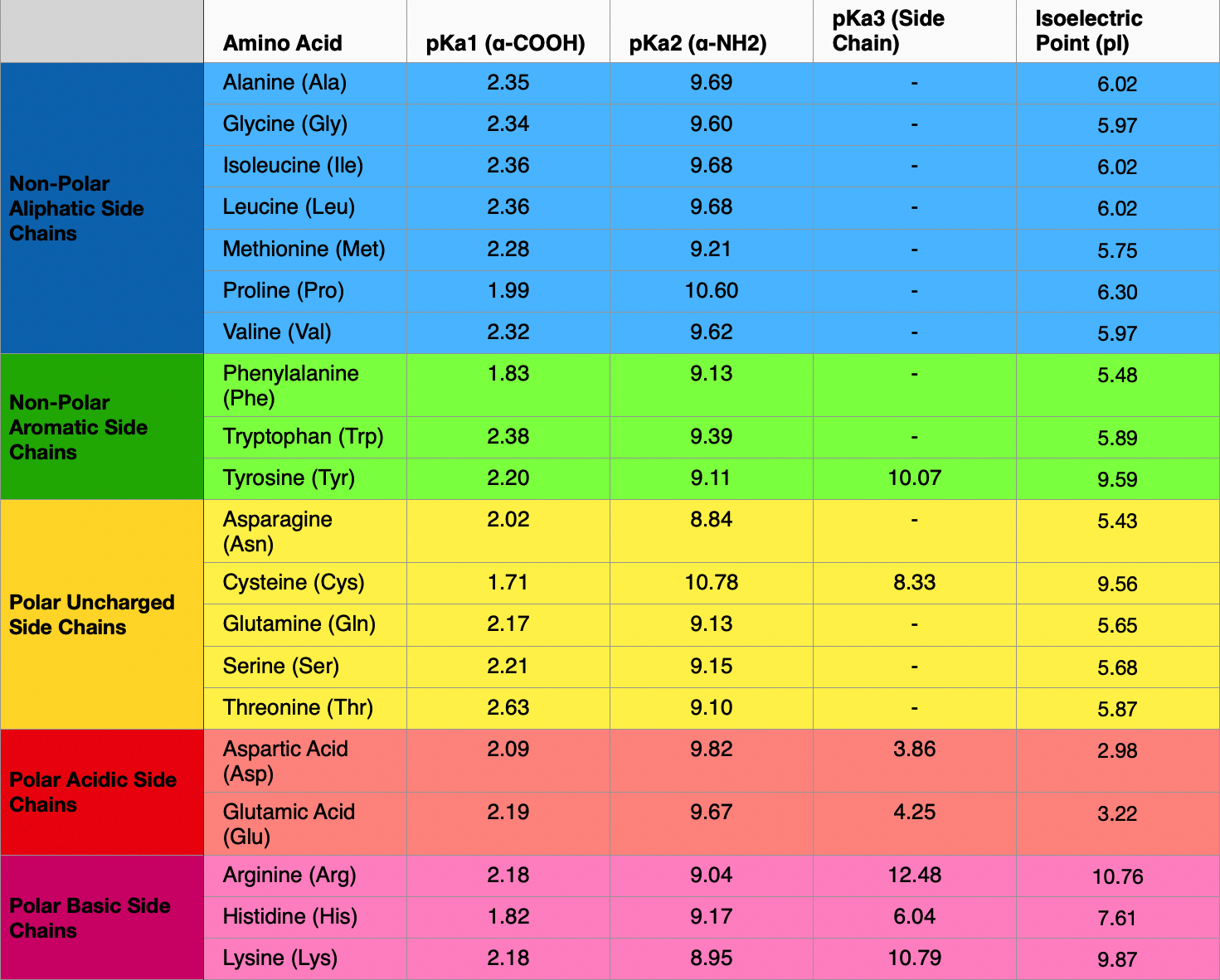
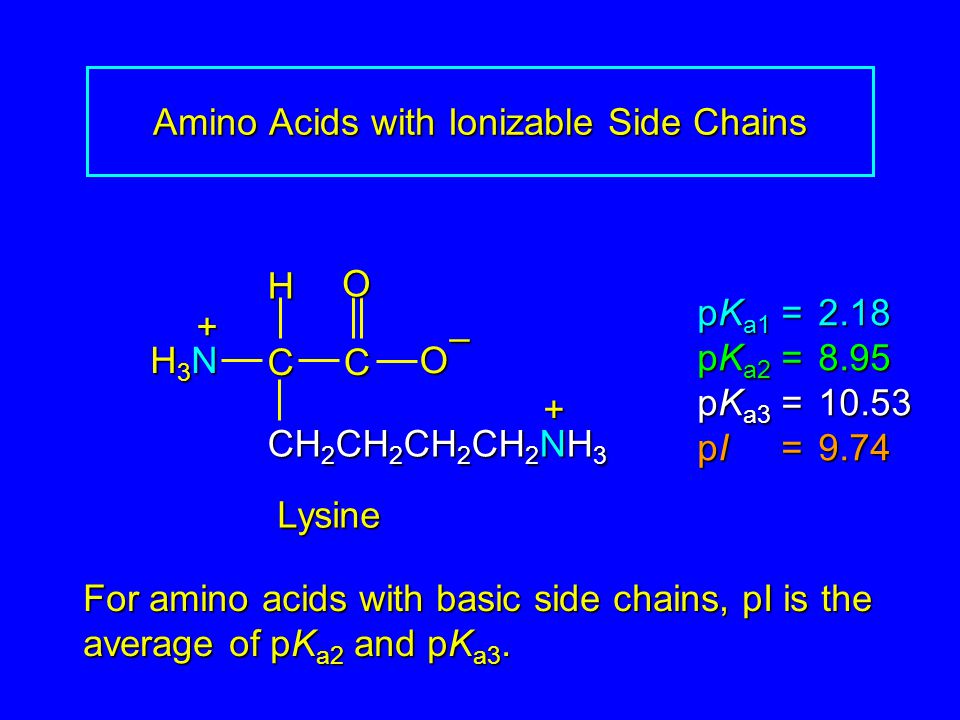
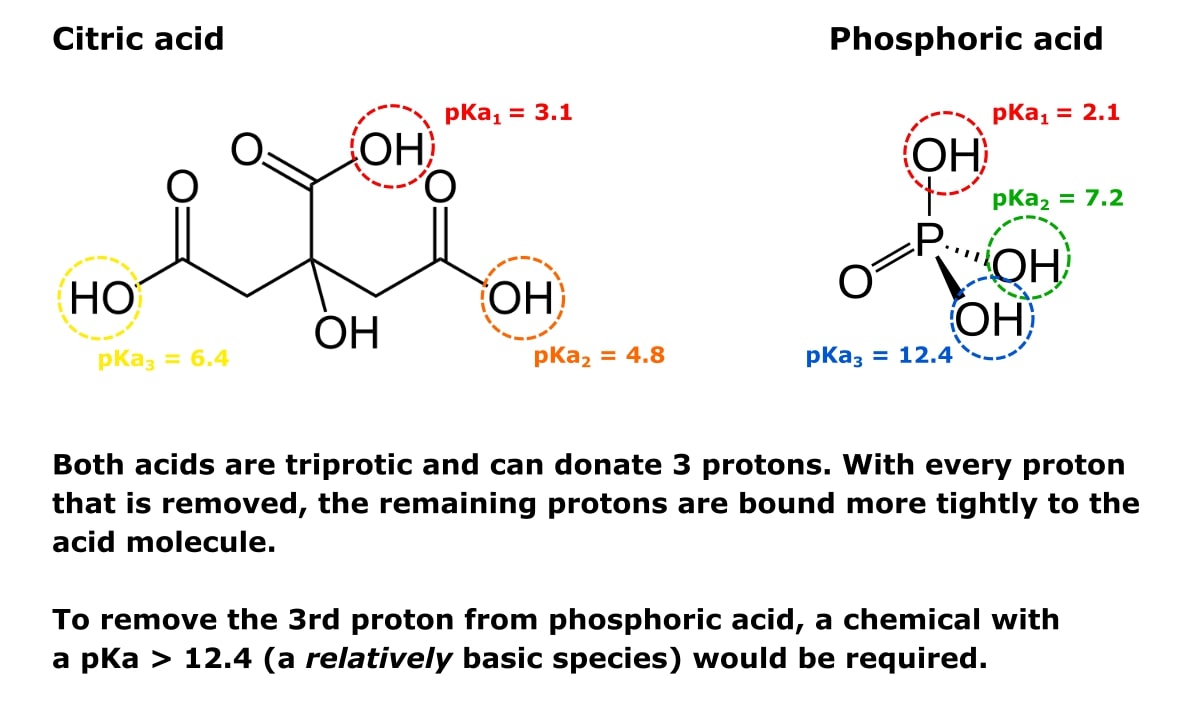
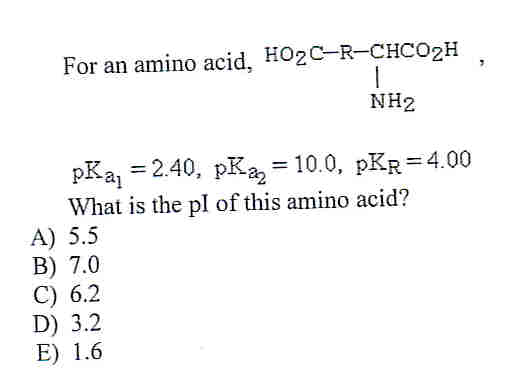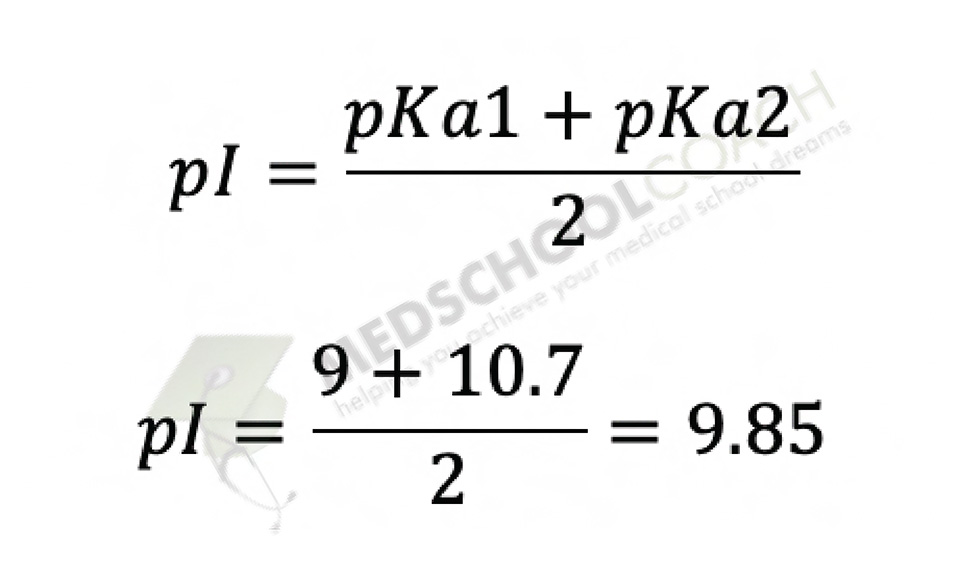Lysine has pKa1 = 2.18, pKa2 = 8.95, pKa3 = 10.53.In which structure lysine will be present at pH = 9.7.

The amino acid methionine has pKa1 = 2.2 and pKa2 = 9.1. If this amino acid is represented by H2L+, what is the major species at pH 6? | Homework.Study.com
How is an isoelectric point calculated in amino acids containing three amino or carboxyl group? - Quora
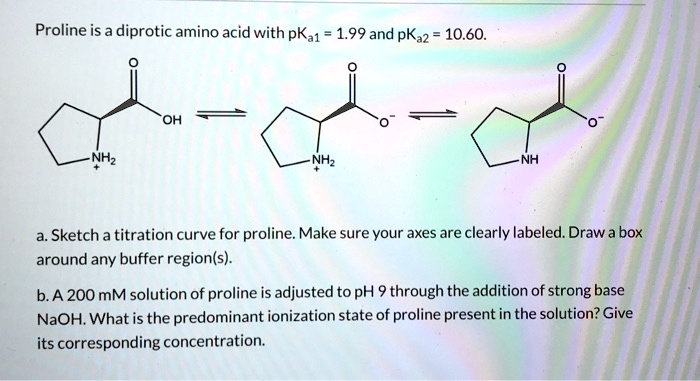
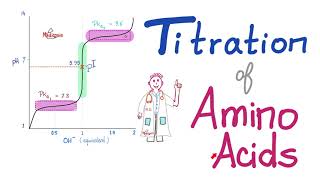
SOLVED: Proline is a diprotic amino acid with pKa1 1.99 and pKa2 10.60. OH NHz NHz Sketch a titration curve for proline. Make sure your axes are clearly labeled Draw a box

Consider the following ionic equilibriumGiven pKa1 =2.3 and pKa2 =9.7Then what is the isoelectric point of alanine?

The amino acid methionine has pKa1 = 2.2 and pKa2 = 9.1. If this amino acid is represented by H2L+, what is the major species at pH 6? | Homework.Study.com