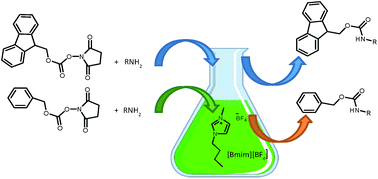
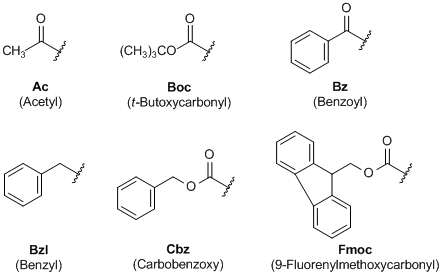
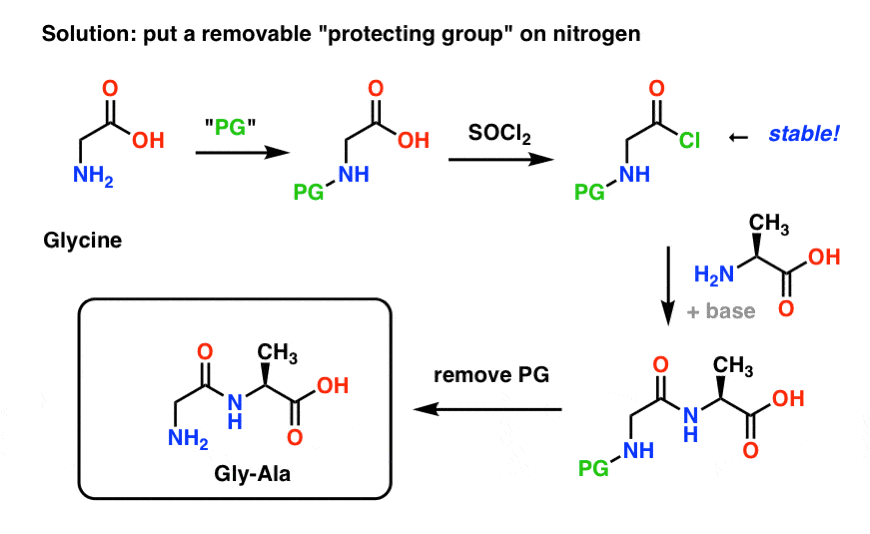
Halofluorination of N-protected α,β-dehydro-α-amino acid esters—A convenient synthesis of α-fluoro-α-amino acid derivatives - ScienceDirect
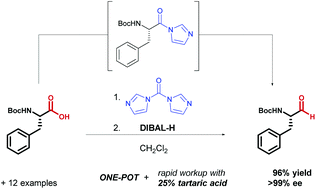
A rapid and efficient one-pot method for the reduction of N-protected α-amino acids to chiral α-amino aldehydes using CDI/DIBAL-H - Organic & Biomolecular Chemistry (RSC Publishing)

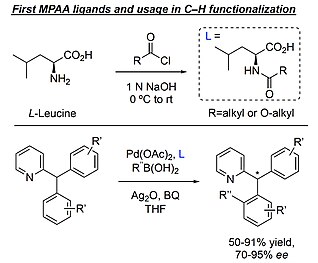
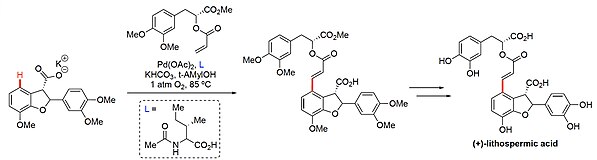
Di-Palladium Complexes are Active Catalysts for Mono-N-Protected Amino Acid Accelerated Enantioselective C-H Functionalization | Catalysis | ChemRxiv | Cambridge Open Engage

Resolution of N-protected amino acid esters using whole cells of Candida parapsilosis ATCC 7330 - ScienceDirect
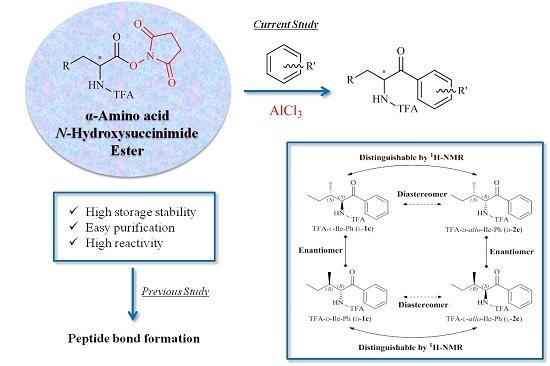
Molecules | Free Full-Text | Synthesis of Chiral TFA-Protected α-Amino Aryl-Ketone Derivatives with Friedel–Crafts Acylation of α-Amino Acid N-Hydroxysuccinimide Ester

Tertiary-butoxycarbonyl (Boc) – A strategic group for N-protection/deprotection in the synthesis of various natural/unnatural N-unprotected aminoacid cyanomethyl esters - ScienceDirect

Direct amidations between doubly N-protected α -phthaloyl amino acids... | Download Scientific Diagram
A Facile Approach to the Synthesis of Benzothiazoles from N-Protected Amino Acids | Russian Journal of Organic Chemistry

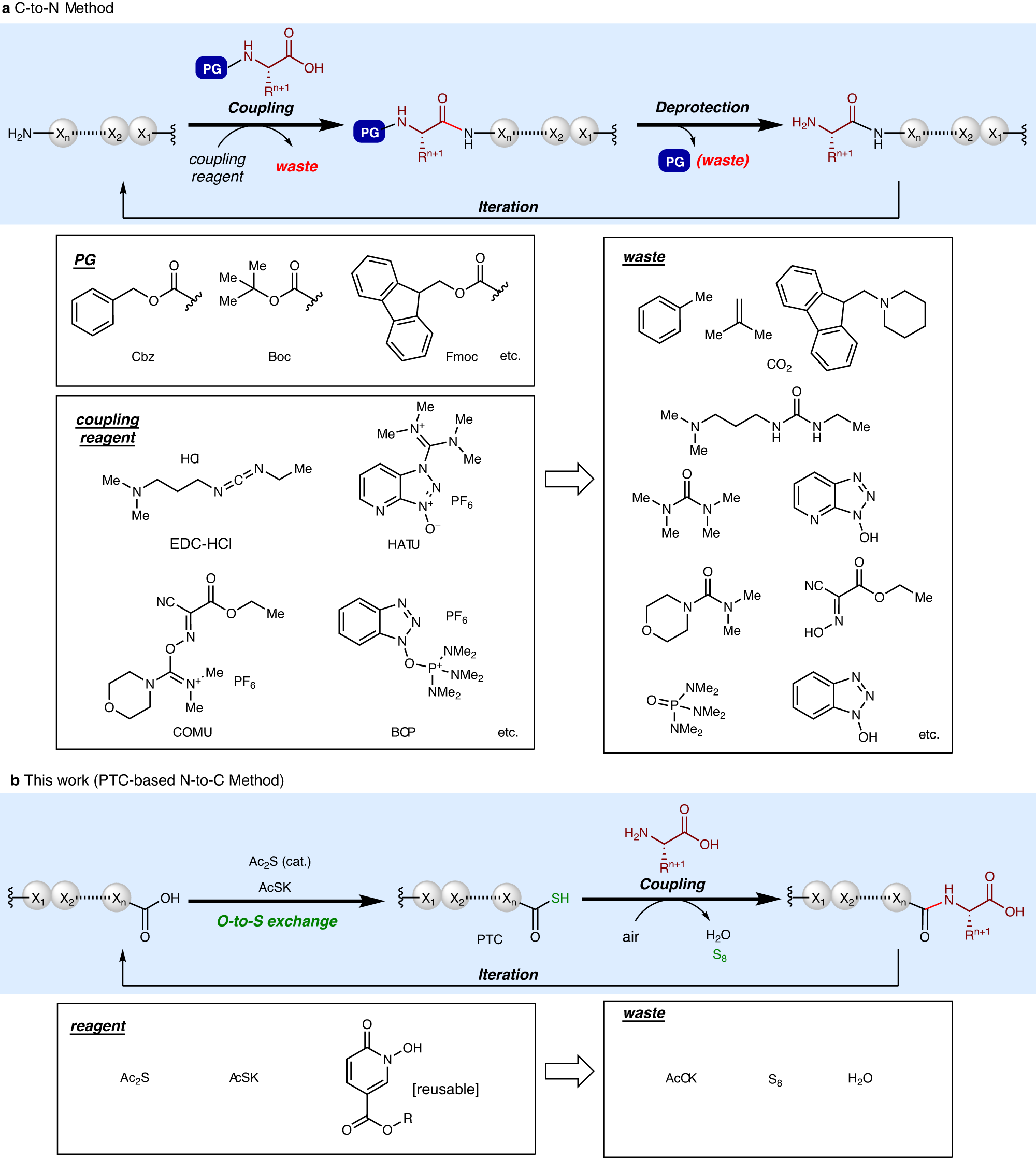
An Efficient Synthesis of Nα-Protected Amino and Peptide Acid Aryl Amides via Iodine-Mediated Oxidative Acylation of Nα-Protected Amino and Peptide Thioacids